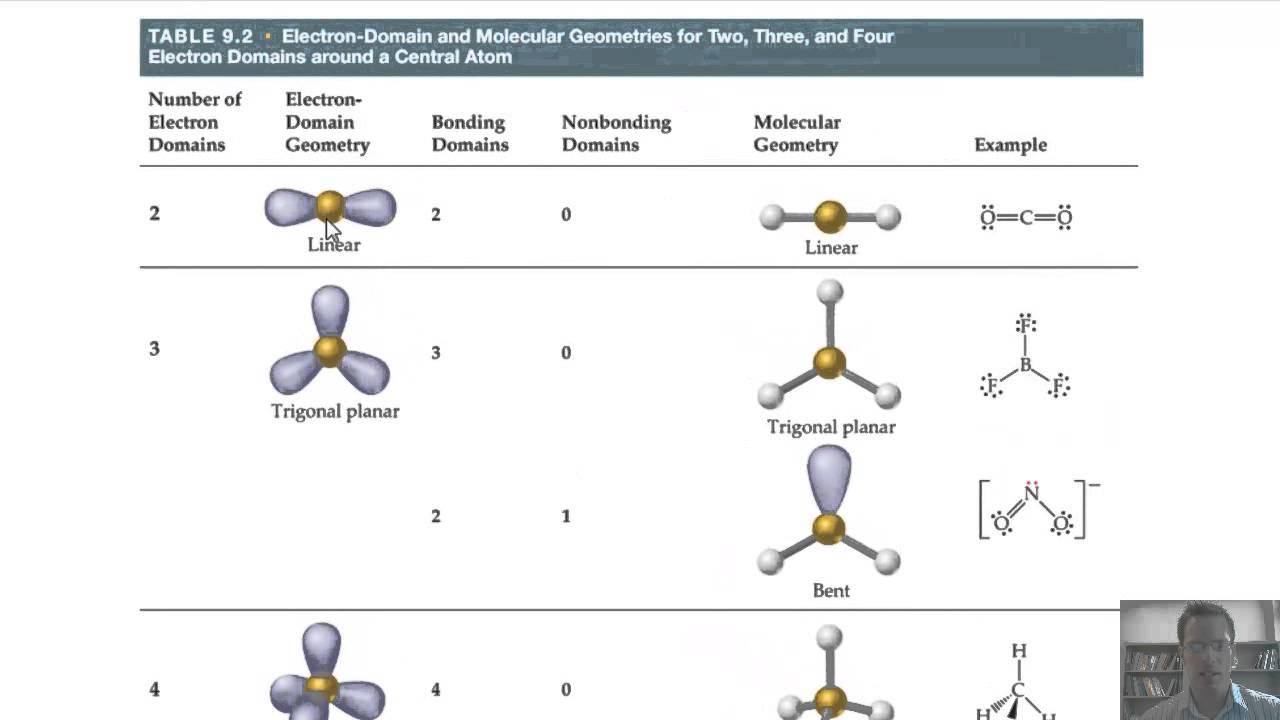 
A in AXE represents the central atom and always has an implied subscript one X represents the number of sigma bonds between the central and outside atoms (multiple covalent bonds-double, triple, etc.- count as one X) and E represents the number of lone electron pairs surrounding the central atom. VSEPR Theory: the Valence Shell Electron Pair Repulsion (VSEPR) model is used to predict the shape of individual molecules based on the extent of electron-pair electrostatic repulsionĪnother way of looking at molecular geometries is through the "AXE method" of electron counting. 
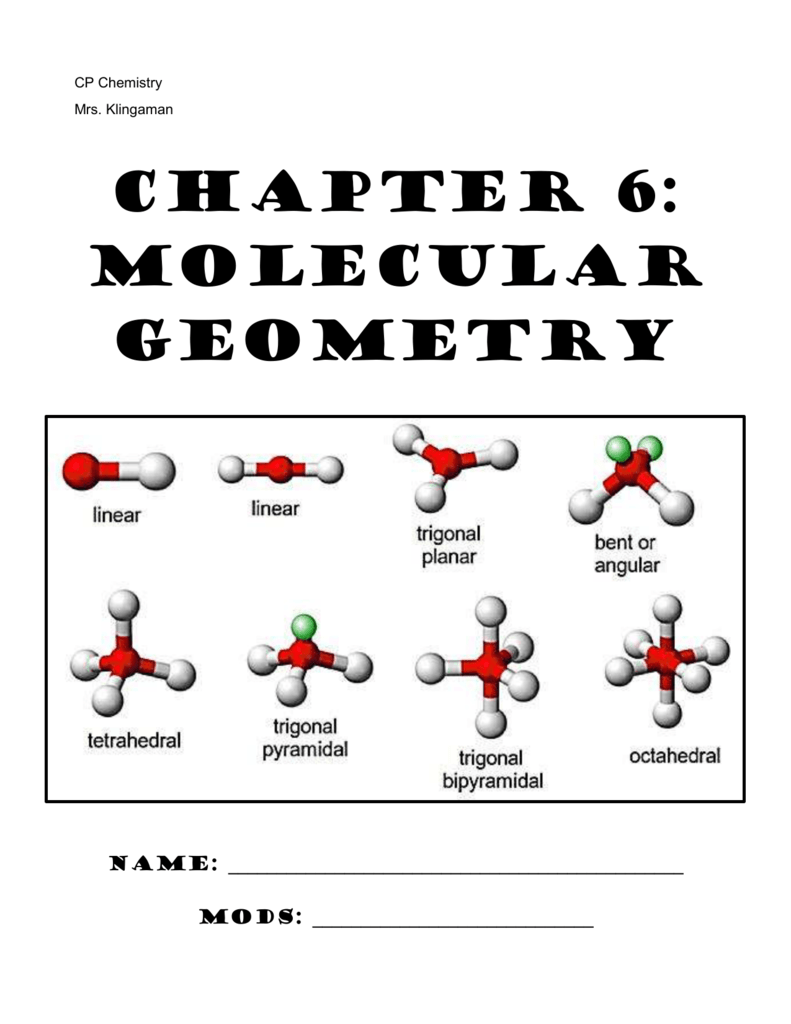
Octahedral: six atoms around the central atom, all with bond angles of 90°.Trigonal bipyramidal: five atoms around the central atom three in a plane with bond angles of 120° and two on opposite ends of the molecule.Tetrahedral: four bonds on one central atom with bond angles of 109.5°.Trigonal planar: triangular and in one plane, with bond angles of 120°.Linear: a simple triatomic molecule of the type AX 2 its two bonding orbitals are 180° apart.The second figure serves as a visual aid for the table. The table of molecular geometries can be found in the first figure. A table of geometries using the VSEPR theory can facilitate drawing and understanding molecules. Molecular geometries (linear, trigonal, tetrahedral, trigonal bipyramidal, and octahedral) are determined by the VSEPR theory. If the central atom possesses partially occupied d-orbitals, it may be able to accommodate five or six electron pairs, forming what is sometimes called an "expanded octet." The orbitals containing the various bonding and non-bonding pairs in the valence shell will extend out from the central atom in directions that minimize their mutual repulsions. If the central atom also contains one or more pairs of non-bonding electrons, these additional regions of negative charge will behave much like those associated with the bonded atoms. The two X atoms (in white) are 180° away from one another. Linear electron geometry: This ball-and-stick model represents a linear compound for formula AX2. An angular separation of 180° places the two bonding orbitals as far away from each other as possible we therefore expect the two chemical bonds to extend in opposite directions, producing a linear molecule. Therefore, the two electron clouds contained in a simple triatomic molecule AX 2 will extend out in opposite directions. The valence shell electron pair repulsion (VSEPR) model focuses on the bonding and nonbonding electron pairs present in the outermost (valence) shell of an atom that connects with two or more other atoms.įundamentally, the VSEPR model theorizes that these regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. VSEPR Theory: a chemistry model used to predict the shape of individual molecules based on electron-pair electrostatic repulsion.The main geometries without lone pair electrons are: linear, trigonal, tetrahedral, trigonal bipyramidal, and octahedral.Molecular geometries take into account the number of atoms and the number of lone pair electrons.Fundamentally, the VSEPR model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
